Learn how cannabis growers select top-performing plants through pheno hunting, from germination to evaluation, traits, and final selection.
Introduction
Pheno hunting (phenotype hunting) is a structured selection process where growers run many seed-derived plants, measure how each expresses key traits, and keep only the winners for cloning, production, or breeding. It is fundamentally an applied genetics problem: the plant’s genotype sets the potential, and the environment (plus genotype × environment interactions) determines what you actually see.
A good pheno hunt looks less like “growing a room” and more like a small experiment: consistent conditions, disciplined tagging, repeatable measurements, and a scoring system aligned to your end goal (commercial yield, craft terpene expression, medical chemotype consistency, outdoor resilience, or other product specs).
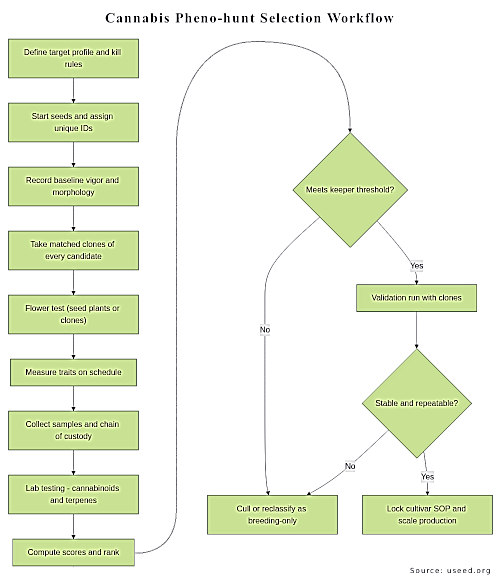
A practical small-to-medium workflow is: (1) start a population from seed, (2) uniquely tag every plant and log actions, (3) take matched clones early, (4) flower the seed plants (or the clones) under controlled conditions, (5) phenotype and sample at defined timepoints, (6) test cannabinoids/terpenes with fit-for-purpose methods, (7) score and select, (8) validate the keepers across at least one repeat run before scaling.
Budgets and timelines are driven by sample counts and testing rigor. Third-party lab testing often ranges from tens of dollars per assay up to several hundred dollars for full compliance panels, while field kits and portable screening tools can reduce per-sample costs but usually reduce accuracy and defensibility.
Finally, selection only matters if you can reproduce the result. That means controlling drift (mother/clone management), confirming chemical consistency with reliable analytics, and documenting decisions for compliance, audits, and IP defensibility.
Definition and genetic background

Pheno hunting is the process of growing a set of plants (usually from seed), observing and measuring how each expresses traits, and preserving the most desirable phenotypes as “keepers,” commonly by keeping clones or a mother plant. In cannabis operations, the traits typically include plant structure, vigor, flowering behavior, yield, aroma, trichome/resin production, cannabinoid profile, terpene profile, and resistance to pests/diseases.
The genotype/phenotype distinction is the backbone of making sense of pheno hunts. A genotype is the genetic makeup (what alleles and variants the plant carries), and the phenotype is the observable expression (morphology, timing, chemistry, performance). Phenotype is shaped by both genotype and environment. In practice, that translates into two operational rules:
First, you cannot evaluate genetics cleanly without controlling the environment. Plant breeding research treats genotype × environment interaction (G×E) as a core reason why “the best” can change with room, season, or management.
Second, many commercially important cannabis traits are partly independent. Morphology does not reliably predict chemotype. Research on terpenoid chemotypes and cannabis diversity emphasizes that chemical phenotype (what cannabinoids/terpenes dominate) can vary independently from visible plant form.
For cannabinoids, there is strong evidence that the THC/CBD ratio (a common chemotype axis) has a substantial genetic basis. Researchers describe three main chemotypes based on THC:CBD ratios (THC-dominant, mixed, CBD-dominant), and segregation patterns in crosses support a major genetic component. Modern genomics adds detail: cannabinoid oxidocyclase genes (including THCAS/CBDAS/CBCAS families) exist in duplicated/clustered genomic regions, and variation and expression in these pathways track with chemical outcomes across lineages and domestication histories.
Terpene expression is similarly measurable and variable. Multiple validated GC–MS approaches exist to quantify cannabis terpenes, and peer-reviewed work shows terpene profiles can differentiate chemovars and change across development and handling.
When and why growers pheno hunt

Growers pheno hunt when they need a plant that performs reliably under their real production constraints, not under a breeder’s ideal conditions. That happens most often when starting from seed for commercial production, when a brand needs a signature aroma/profile, when a medical program needs chemical consistency, or when moving genetics into a new environment (new facility, new region, new cultivation style).
Pheno hunts are also a response to market and compliance realities. In regulated markets, potency and labeling accuracy are scrutinized, and published evidence shows that labeled THC potency can be inflated or inconsistent in retail contexts. That creates pressure to use robust sampling and credible analytics so your selection decisions hold up outside your own room.
A practical way to decide whether a pheno hunt is “worth it” is to quantify uncertainty and downside:
If you run un-hunted seed lots into production, you accept variability in yield, morphology, harvest timing, and chemistry. That variability can translate into inconsistent product specs and higher QA rejection risk.
If you pheno hunt, you spend time and money up front (space, labor, testing) to reduce variability later. In quality systems, this is the same logic as qualifying a supplier or validating a process: invest early so later lots are predictable.
As for “when” in the crop cycle: pheno hunting is most efficient when you plan for it at the start, because the biggest operational failure is discovering you cannot identify, reproduce, or legally document the winning plant after harvest. The standard industry description of a seed pheno hunt explicitly includes taking matched clones and marking everything before flowering.
Practical protocol for a small-to-medium pheno hunt

This protocol is written for authorized cultivation and breeding programs. Regulations vary widely; design your SOPs to meet local licensing, security, tracking, and testing rules.
Scale targets (small-to-medium): 20–200 seeds per “hunt batch” is common because it balances genetic diversity with manageable labor and testing costs. The right number depends on how rare the target trait is and how stable the seed line is.
Core principle: treat the hunt as a controlled comparison. Keep the environment consistent, randomize plant positions, and record deviations (equipment failures, pest incidents, irrigation changes). That reduces the risk that your “winner” is simply the plant that got the best corner of the room.
Tagging and records: Good Agricultural and Collection Practice frameworks for medicinal plants emphasize documentation and traceability, including batch records and retention. The same discipline is what makes a pheno hunt defensible.
Step-by-step workflow
Pre-hunt setup (before you germinate)
Define: (1) target product profile, (2) trait weights, (3) minimum thresholds (cull rules), (4) sampling plan, (5) lab methods you will use, and (6) how you will preserve winners (clones/mothers/tissue culture).
Set up a simple data model that you can maintain:
- Unique Plant ID (barcode/RFID optional, waterproof label required)
- Genetics source/lot, sow date
- Location history (room/rack position over time)
- Key events (clone date, flower induction date, harvest date)
- Observations (with dates and who measured)
- Sample chain-of-custody and lab results (linked to Plant ID)
Seedling stage (days to early weeks)
Keep conditions stable and avoid early stress tests unless stress tolerance is the goal; early stress can confound later trait expression and makes comparisons noisy.
Tag each plant immediately. If a label can fall off, assume it will. Use redundant ID methods (tag + pot label + map + photo of ID in frame). Traceability guidance in U.S. frameworks is explicit about early labeling and batch assignment.
Vegetative growth and clone capture (early to mid veg)
Take at least one matched clone per seed plant early enough that clones can be rooted and held while the seed plant is flower-tested. This “matched clone” approach is necessary for pheno-hunt workflows because it preserves the genotype of each candidate.
If your operation can support it, take multiple clones (e.g., 2–4) per candidate so you can run a small replicate or rerun quickly if one fails. Replication is the standard way plant breeding reduces random environmental noise.
Flowering trial (phenotyping window)
Flower induction and flowering duration vary by genotype and photoperiod response. Controlled-environment studies show cannabis flowering can initiate under different daylength thresholds depending on genotype, and cultivars can differ in response above “classic” schedules.
Plan to measure traits at defined timepoints, not just when you remember. Chemistry, especially cannabinoids and terpenes, changes across floral development. A controlled study tracking chemotypes weekly demonstrates measurable time-course changes and uses replicated clones to separate plant-to-plant variation from developmental effects.
Harvest, dry/cure, and post-harvest phenotyping
Sampling method matters because potency and terpene results are sensitive to where and how you sample, how you homogenize material, and how your lab prepares samples. Standards groups have proposed sampling guidance for post-harvest materials to improve representativeness.
If your goal includes marketable flower quality, incorporate at least one structured sensory evaluation (aroma intensity, note descriptors, harshness, visual appeal), with panel bias controls (blind IDs, consistent lighting, consistent grind method) and written descriptors. Research and analytical commentary increasingly emphasize that aroma quality is not fully explained by a single terpene list, which strengthens the case for combining sensory and instrument data.
Traits to evaluate and how to measure them
Use traits that tie directly to your product spec and production constraints. Cannabis morphology and development can be characterized systematically (leaf morphology, plant architecture, flowering development, trichome features), and published phenotyping work shows that a broad trait set can be measured consistently across stages.
A practical small-to-medium trait set:
- Morphology and structure: internode length, branching pattern, canopy shape, lodging risk, trim labor estimate. Biomass yield can be associated with architecture traits in hemp hybrids, supporting the idea that structure metrics can be predictive.
- Vigor and uniformity: early growth rate, stress recovery, root vigor (via clones), uniformity across clones (stability indicator).
- Flowering behavior: days to initiation, days to harvest maturity, uniform ripening. Photoperiod response is cultivar-dependent in controlled experiments, so document your photoperiod and cultivar response rather than assuming one schedule fits all.
- Yield: dry flower yield, marketable fraction, density/structure, harvest index.
- Cannabinoid profile: total cannabinoids and the key ratios relevant to your market (examples: THC-dominant, CBD-dominant, balanced types; minor cannabinoids depending on product plan). Classical inheritance and chemotype classification literature supports treating THC/CBD ratios as a key selection axis.
- Terpene/volatile profile: total terpene content (if relevant), dominant terpenes, and repeatability across runs; GC–MS methods are widely used and validated for terpene quantification in cannabis matrices.
- Pest/disease resistance: visible symptoms, incidence under controlled exposure history, and survival/quality under disease pressure. Powdery mildew resistance is an active research area with mapped resistance loci in cannabis, supporting selection based on observed resistance.
- Stability (repeatability): the same clone should hit a similar chemical and performance target across at least two runs or at least two environments. Plant breeding treats this as a multi-environment or multi-run validation problem because G×E can change rankings.
Sample pheno hunt schedule
This is a representative schedule for a photoperiod-type hunt where you take clones, flower-test candidates, then validate the top selections in a second run. Flower initiation and duration vary by cultivar, so treat the weeks as planning ranges, not guarantees.
| Week range | Stage | What you do | What you record | Decision point |
|---|---|---|---|---|
| 0–1 | Start | Germinate/establish seedlings; assign unique Plant IDs | Plant ID, lot/source, start date, initial vigor rating | Cull obvious weak/non-viable seedlings |
| 2–4 | Early veg | Standardize training/spacing; start photo log | Height, node count, branching notes, vigor score | Early cull for severe structural issues |
| 3–5 | Clone capture | Take matched clones from every candidate; label clones with parent Plant ID | Clone date, clone success rate per plant | Remove plants that cannot be cloned reliably |
| 5–6 | Pre-flower | Finalize shortlist if space-limited; randomize positions | Final candidate list, room map | Lock candidate count for flower trial |
| 6–14 | Flower trial | Run flower test; score weekly | Flower initiation date, weekly morphology/aroma notes, pest/disease notes | Mid-trial cull rules (disease, herm traits, severe instability) |
| 12–15 | Harvest & dry | Harvest by defined maturity criteria; dry/cure consistently | Harvest date, wet/dry weights, bud structure/photos | Select plants for lab sampling |
| 14–16 | Testing | Send samples for cannabinoid/terpene tests; log chain-of-custody | Sample IDs linked to Plant IDs; lab methods and results | Compute selection scores; pick top tier |
| 16–24 | Validation run | Grow clones of top tier under production-like conditions | Repeat the same metrics; compare to first run | Confirm keepers; discard inconsistent winners |
Sampling and lab testing options

Testing strategy should match the decision you’re making. If you are choosing between “keep and clone” candidates, you need precision and comparability more than you need a compliance certificate. If you are qualifying a cultivar for regulated product release, you need defensible sampling and accredited lab methods.
Cannabinoid testing
Most potency testing in cannabis and hemp relies on chromatographic methods such as HPLC (often with UV/PDA detection) because it can quantify acidic and neutral cannabinoids without forcing decarboxylation, which is important for accurate total THC calculations. Industry method development and official-method ecosystems exist, including official methods and proficiency testing programs.
Quality control is moving toward better harmonization via reference materials and proficiency testing. National Institute of Standards and Technology issued RM 8210 as a hemp plant reference material intended to help harmonize cannabinoid and toxic element measurements. AOAC International runs cannabis/hemp proficiency testing and the Cannabis Analytical Science Program to improve method performance and comparability.
Terpene and volatile testing
Terpene profiling is typically performed using GC-based methods (GC–FID and/or GC–MS), including headspace approaches, and multiple peer-reviewed methods report validated quantification for key terpene panels in cannabis matrices. Because terpene content and cannabinoid/terpene ratios can shift during plant development and post-harvest handling, define your sampling stage and handling SOP before you start sending samples.
Quick field screening options
Field screening is useful for triage: narrowing 200 candidates to 40 before you pay for full lab panels. It is not a substitute when you need defensible numbers.
- TLC/HPTLC kits: Thin-layer chromatography approaches are widely used in cannabis analysis contexts as lower-cost screening tools, and published work supports TLC/HPTLC as a practical identification/semiquant approach when properly validated. Commercial kits can be low cost per test (single-digit currency units in some offerings), but your accuracy depends heavily on operator skill, calibration, and SOP discipline.
- Portable NIR: Peer-reviewed studies show handheld near-infrared devices can predict major cannabinoids in cannabis/hemp inflorescences with chemometric models, making them useful for rapid, non-destructive screening—if you invest in calibration for your genetics and matrix.
- Lab variability reality check: Even accredited labs can disagree on totals if sample prep and methods are inconsistent, which is why harmonization tools (reference materials, PT) matter when you compare candidates across time and labs.
Selection metrics and scoring systems

A scoring system prevents two classic failure modes: (1) choosing with your memory, and (2) choosing for the wrong goal (e.g., picking the loudest aroma when your buyer needs 8-week harvest consistency). Multi-trait selection is standard practice in breeding and varietal evaluation, and the same logic applies to pheno hunting.
Example scoring rubric
This rubric yields a 100-point index. It assumes you are selecting a production cultivar (not strictly a breeding parent). Adjust weights to your business model and compliance constraints.
Hard cull rules (fail = stop scoring):
- Cannot be reliably cloned / maintained as a cultivar
- Severe disease susceptibility under your standard IPM conditions
- Chemical profile outside your required spec window (for example, wrong THC:CBD class for the product)
Scoring table:
| Domain | What you measure | Points | Example thresholds |
|---|---|---|---|
| Structure & morphology | Architecture, ease of training/harvest, trim labor estimate | 0–15 | Keep ≥10 for production efficiency 23 |
| Vigor & uniformity | Growth rate, uniformity across clones, stress recovery | 0–15 | Keep ≥10; uniformity matters for scaling 8 |
| Flowering timing | Days to flower initiation + harvest window | 0–10 | Keep ≥6 to meet scheduling needs 19 |
| Yield & marketable fraction | Dry yield + usable fraction | 0–20 | Keep ≥14 for commercial yield focus 23 |
| Cannabinoid profile | Potency + ratios by target (lab or validated screening) | 0–20 | Keep ≥16 and within chemotype spec 10 |
| Terpene/aroma quality | Sensory score + terpene panel | 0–15 | Keep ≥11 for craft focus 12 |
| Pest/disease resistance | Incidence/severity, survival, quality impact | 0–5 | Keep ≥3; weight higher outdoors 24 |
Decision thresholds:
- 80–100: keeper tier (advance to validation run)
- 70–79: “maybe” tier (advance only if you need more options or a specific niche trait)
- <70: cull for production, consider only as breeding material if it carries a rare trait
Flowchart infographic: selection workflow
Breeding and stabilization after you find a winner
A keeper phenotype can be used in two main ways: as a cultivar (clone-propagated for consistent production) or as a parent (to create seed lines that reproduce desired traits). Which path you choose changes what “best” means.
Guide: The Ultimate Beginner’s Guide to Breeding Cannabis: From Seed to Strain
If you are keeping it as a production cultivar
Your stabilization goal is operational, not genetic: maintain a healthy, true-to-type clone line. That means:
- Protecting identity and traceability (mother/clone ID discipline)
- Confirming chemical and performance repeatability across at least one validation run
- Managing sampling and testing defensibly because potency and labeling accuracy are high-scrutiny areas in many markets
If you are breeding it to stabilize traits in seed
Breeding adds another layer: you want descendants that repeatedly express the target phenotype, not just the original plant.
Two core methods the cannabis industry often borrows from classical plant breeding language:
- Backcrossing (BX): repeatedly crossing offspring back to a recurrent parent to recover most of the recurrent parent genome while introducing or fixing a specific trait from a donor. Plant breeding references define backcrossing this way and describe its use for trait introgression and defect correction.
- Selfing (S1/S2…): repeated self-fertilization increases homozygosity across generations, which can make trait expression more uniform, though it can also expose deleterious recessives and reduce vigor depending on species and starting heterozygosity.
A realistic stabilization plan usually takes multiple selection cycles. Each cycle is: make a population, phenotype it under controlled conditions, test chemistry, select the best individuals, then repeat. This is the same logic used in multi-generation selection and stability work across crop breeding, where multi-environment or multi-run testing is used to confirm performance.
For cannabinoid chemotypes, major genetic determinants exist, but modern genomic work shows gene clusters, duplications, and expression differences that can complicate simple “single marker = outcome” assumptions across diverse germplasm. That is a strong argument for combining genotype information (if you have it) with measured chemotype outcomes over generations.
Common pitfalls and troubleshooting
Pitfall: losing plant identity. One unlabeled clone tray can ruin months of work. Redundancy in labeling and record retention is emphasized in traceability guidance.
Pitfall: selecting on one run. Many traits are sensitive to environment and G×E. A “winner” can fail on repeat. Validate across runs before scaling.
Pitfall: comparing lab results that are not comparable. Variability across labs and methods is documented in hemp/cannabis analytical contexts, which can mis-rank candidates if you switch labs or change prep. Use consistent labs and, when possible, harmonization tools like reference materials and PT participation.
Pitfall: sampling bias. Cannabinoid content can vary within the plant and across material strata, so inconsistent sampling can create “phantom winners.”
Pitfall: confusing aroma “loudness” with market fit. Analytical work and industry discussion increasingly note that aroma quality is multi-factor and not fully captured by a simple terpene list, so combine sensory evaluation with GC-based profiling when aroma is central.

